|
Wiener focused on the mathematical models of Brownian motion and other stochastic processes. In the early 1900s, Norbert Wiener was also an important figure in learning about Brownian motion. 
And for his work, Perrin was awarded the Nobel Prize in physics in 1926. Jean Baptiste Perrin then furthered Einstein’s studies. And in 1905 he published the first paper on the topic. From this observation, he deduced there was some other phenomenon occurring.įollowing Brown’s discovery, Einstein continued investigating this phenomenon. He was examining plant seeds and was puzzled by their movement even when they were dead. The Scottish botanist Robert Brown was the first one known to investigate Brownian motion in 1827. The beads each have a random trajectory and do not all move in the same direction. The short movie above shows fluorescent beads moving through a solution. 
Fluorescent beads can be seen moving in solution. The individual particles are tracked by detecting the photons or light released by single molecules as they move through the solution. Particles move away from their original position and randomly distribute.Ī common experiment where Brownian movement can easily be tracked is watching fluorescent dyes in a solution. All particles in a liquid or gas are moving due to Brownian motion.ĭiffusion happens in part due to Brownian motion. Examples of Brownian Motionīrownian motion can be hard to observe. The effect of all the smaller particles hitting the larger particles is enough to counteract gravity and cause the large particles to stay in solution. Instead, Brownian movement is what keeps the particles in solution. The larger particles stay suspended in solution and do not settle out to the bottom as predicted due to gravity. Milk and fog are common examples of colloids. Therefore, smaller particles will move faster and travel further than larger particles.Ī colloid is a homogeneous mixture with large particles suspended in a solution of another substance. Smaller Particles: particle speed is inversely related to the size of the particle.The same is true on the smaller scale with individual particles. The water will be easier to move through. Think about trying to move through molasses (very viscous) compared to water (less viscous). Less Viscous Solution: The more viscous a solution is the more energy it takes for a particle to move through it.Increase Temperature: The higher the temperature, the more energy each particle has.Factors that Increase Motion of Particles As the temperature increases, there is more energy in the system, and motion increases. Brownian motion is also called thermal noise because of its relationship to temperature. Other names for Brownian motion include Brownian movement and pedesis (Greek for ‘leaping’). The orange line is after the most amount of time. The blue line is after the least amount of time has passed. Probability of finding a particle at a certain distance after different amounts of time. This means that as time goes on, the particle is more likely to be further away from its starting location. The distance of a particle from its starting position will be a Gaussian distribution, with the width of the Gaussian increasing over time. Brownian movement is often modeled using a ‘ random walk’. Each step is random and independent of the previous step. The steps the particle takes are non-correlated. Yellow particles collide randomly with black particles and their path is tracked in blue. Particle one hitting particle two will cause both particles to shift their momentum(direction and speed). Similar to how billiard balls hitting cause them each to change direction, the same is true of molecules. Instead, the movement occurs because of particles colliding with each other in a liquid or gas. 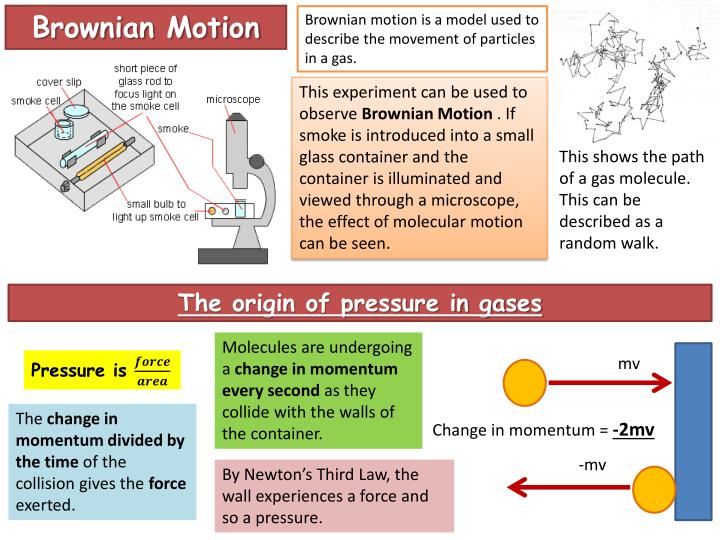
Particles are never staying completely still. This movement occurs even if no external forces applied. Brownian motion is the random movement of particles in a liquid or gas.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
